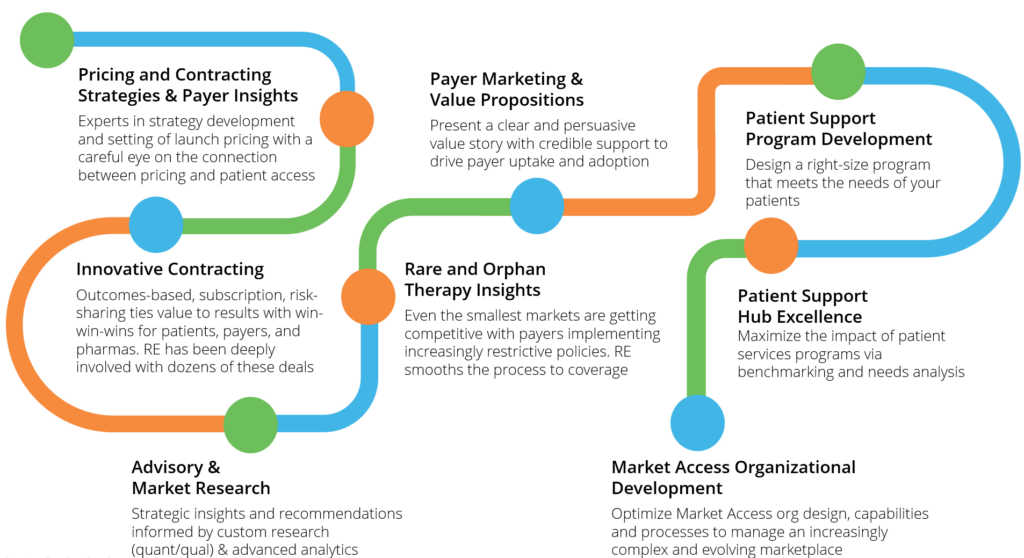
Solutions
Real Endpoints brings a wide range of strategic and operational market access solutions to our clients.
Platforms
Real Endpoints offers platforms to help our clients maximize the potential of their market access teams.
Solutions
News and Events

RE at the 2026 J. P. Morgan Healthcare Conference
by Real Endpoints
Real Endpoints will be meeting at JP Morgan in January with a variety of folks on these and other issues – and would love to sit down with you as well. We can fill you in on what we see as the implications of policy and payer changes on coverage, reimbursement, and patient support; what we’re expecting for 2026; and discuss the specific topics you’re most concerned about.
December 5, 2025
news >
Real Endpoints board member Michael Sherman spells out the implications of recent Massachusetts-based GLP-1 coverage decisions for different healthcare stakeholders.
by Real Endpoints
Real Endpoints board member Michael Sherman spells out the implications of recent Massachusetts-based GLP-1 coverage decisions for different healthcare stakeholders. Thing is these implications could also easily apply to any number of drugs in other competitive disease spaces, where there is high budget impact and more than a little uncertainty about clinical benefit.
June 7, 2025
LinkedIn >
Webinar: Designing Patient Support Services for Impact: Best Practices
Susan Raiola, President, Real Endpoints
Rob Holmes, VP, Patient Experience & Reimbursement, GSK
Ilias Kifnidis, Head of Patient Services, Genmab
April 16, 2026
11am ET
