Access Meter measures the access challenges for a given drug – systematically and quantitatively. Based on an objective understanding of payer decision-making, it guides critical investment and strategy choices by showing which access challenges a new drug will face and how severe they’ll be.
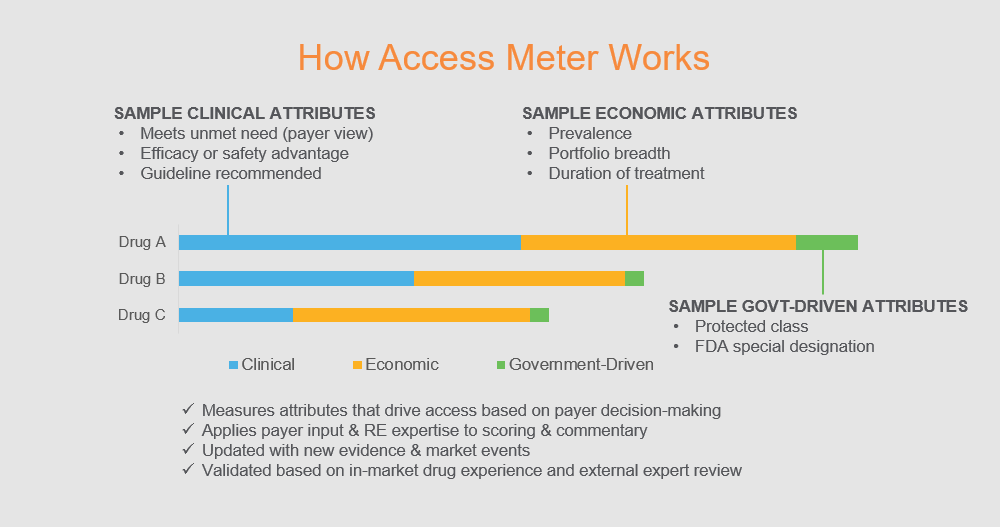
100% focused on market access
Guide crucial company decisions
Range of Access Meter Options
Recently Scored Drugs
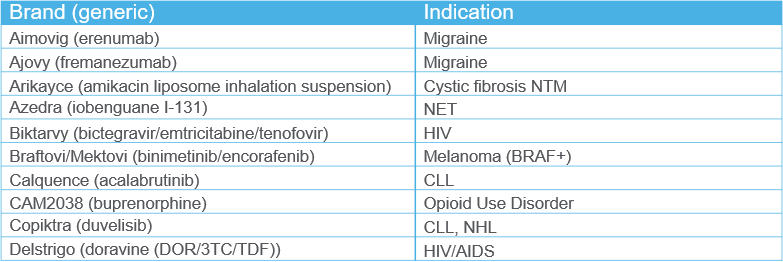
Click here to view the complete list of drugs.

Sage Therapeutics’ brexanolone, a first-in-class GABA-A modulator for treating postpartum depression, successfully addresses a number of key access criteria – in particular, a high unmet need (it would be the first approved drug for treating postpartum depression) – and a very limited duration of therapy, a single infusion, which means that payers won’t be paying for the drug year-in and year-out. The drug wins additional points for getting FDA breakthrough status and a few more as medical-benefit drug – a status which makes controlling access more difficult for payers. And Sage will also benefit from strong support by effective women’s health patient advocacy groups – payers will be wary of the negative press these groups can generate……………………………..To continue reading click here